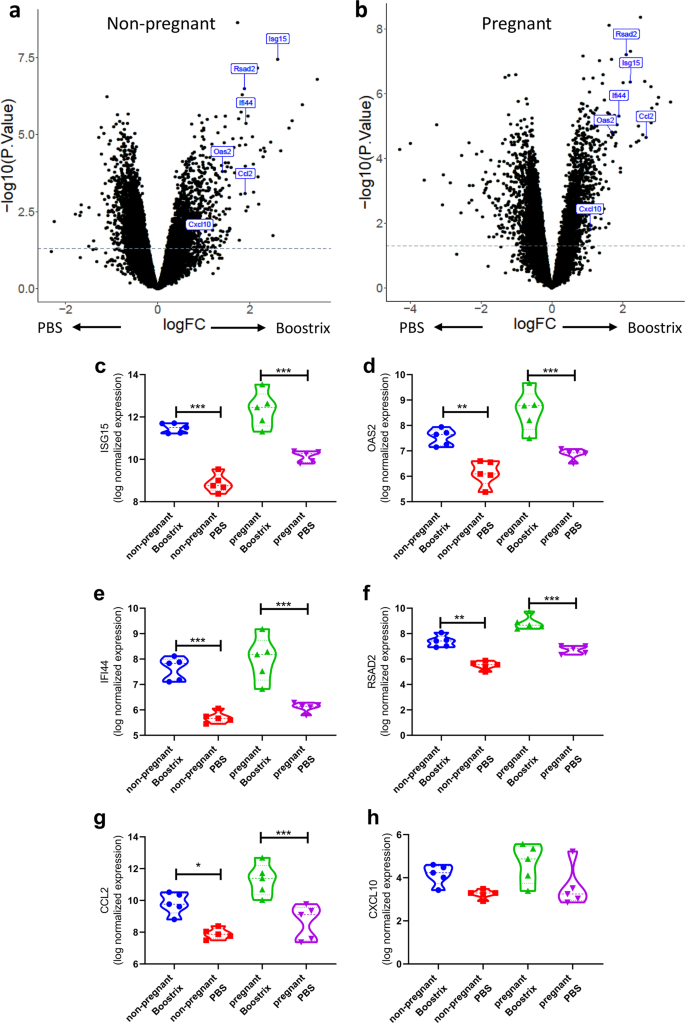
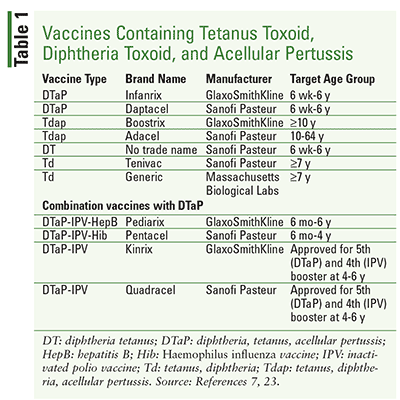
Boostrix Injection Uses In Pregnancy
The whooping cough vaccine is usually given to pregnant women at 28 weeks can be given anytime between 20 32 weeks of each pregnancy and should be given as early as possible from 20 weeks to women who have been identified as being at high risk of early delivery.
Boostrix injection uses in pregnancy. The vaccine does not contain any live bacteria or viruses and cannot cause any of the diseases it protects against. Boostrix similar to boostrix ipv but without the polio component is one of the vaccines routinely recommended in the us for immunisation of pregnant women. The vaccine offered to pregnant women in the uk is called boostrix ipv. This is also used as a pre school booster vaccine and protects against diphtheria tetanus and polio as well as pertussis.
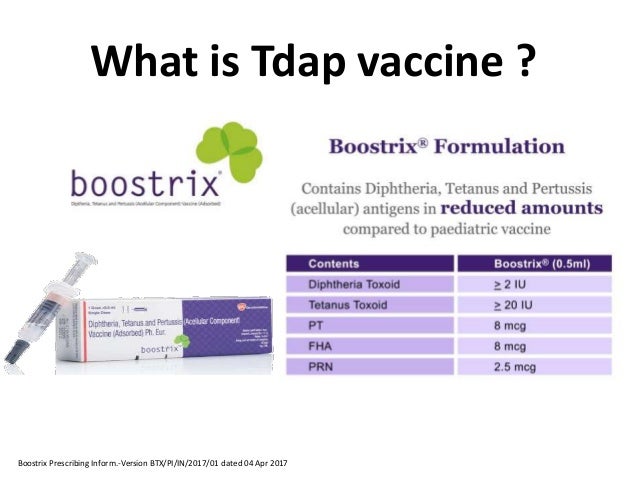
Manufacturer pregnancy registries both manufacturers of tdap vaccine sanofi pasteur for adacel and glaxosmithkline for boostrix created pregnancy registries to collect information from pregnant women who got tdap vaccine. Learn about age restrictions side effects use during pregnancy and more. Boostrix is a vaccine for the prevention of tetanus diphtheria and pertussis. Vaccination during pregnancy preferably at 20 32 weeks means your body produces antibodies that get passed on to your baby before birth.
Influenza vaccine is also free for pregnant women and recommended at any time during every pregnancy. There have been no reported safety concerns in the us with the use of the vaccine in pregnancy. However you may need boostrix vaccine during pregnancy to protect your newborn baby from pertussis. Healthcare providers are encouraged to register women by calling 1 888 452 9622.
Your doctor should determine whether you need boostrix during pregnancy. Use in specific populations pregnancy. Young babies are most at risk for severe life threatening complications from pertussis. To date vaers has not found any safety signals among pregnant women or their babies after tdap vaccination.
If you are pregnant your name may be listed on a pregnancy registry. During pregnancy the immunity developed by a mother after vaccination during pregnancy is passed on to her baby in the womb. Boostrix or adacel can be co administered with influenza vaccine to pregnant women. From 1 july 2019 this booster dose is funded from the second trimester of every pregnancy and recommended to be administered from 16 weeks preferably within the second trimester.
This immunity helps protect the baby during the first few months of life.
Https Encrypted Tbn0 Gstatic Com Images Q Tbn 3aand9gcrzeyeowavbrr 2x9aa34ehoz Onw8jq0pcrmh Uoc Usqp Cau

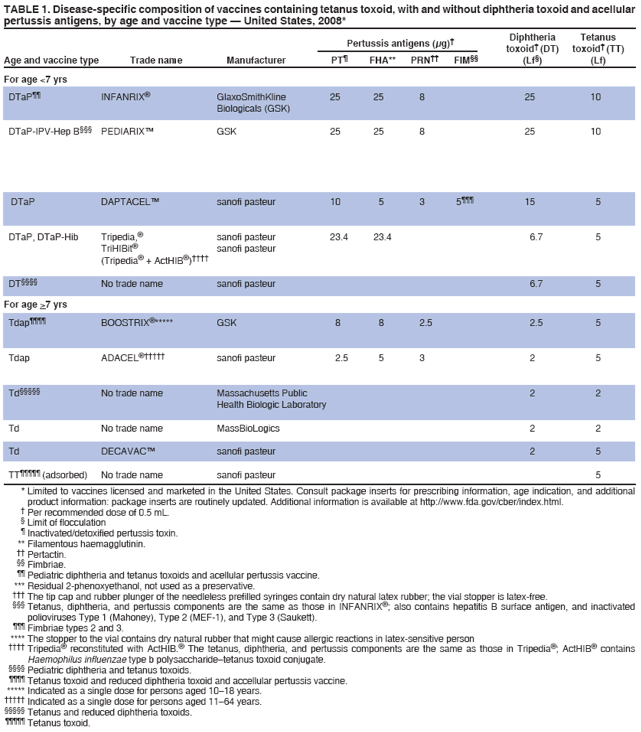














































/doctor-giving-patient-injection-in-volunteer-clinic-573103329-5b3a6ad2c9e77c001a4b70d5.jpg)